Abstract
Cervical spondylotic myelopathy (CSM) is a neurologic condition that develops insidiously over time as degenerative changes of the spine result in compression of the cord and nearby structures. It is the most common form of spinal cord injury in adults; yet, its diagnosis is often delayed. The purpose of this article is to review the pathophysiology, natural history, diagnosis, and management of CSM with a focus on the recommended timeline for physicians suspecting CSM to refer patients to a spine surgeon. Various processes underlie spondylotic changes of the canal and are separated into static and dynamic factors. Not all patients with evidence of cord compression will present with symptoms, and the progression of disease varies by patient. The hallmark symptoms of CSM include decreased hand dexterity and gait instability as well as sensory and motor dysfunction. magnetic resonance imaging is the imaging modality of choice in patients with suspected CSM, but computed tomography myelography may be used in patients with contraindications. Patients with mild CSM may be treated surgically or nonoperatively, whereas those with moderate-severe disease are treated operatively. Due to the long-term disability that may result from a delay in diagnosis and management, prompt referral to a spine surgeon is recommended for any patient suspected of having CSM. This review provides information and guidelines for practitioners to develop an actionable awareness of CSM.
Introduction
Cervical spondylotic myelopathy (CSM) is the most common form of spinal cord injury in adults, accounting for 54% of nontraumatic spinal cord injury in North America.1⇓–3 Cervical spondylosis is a progressive disease defined by degenerative changes affecting the vertebrae, intervertebral disks, facets, and associated ligaments. These changes precipitate CSM through direct compression of the spinal cord and/or surrounding blood vessels.1 This disease can potentially produce long-term disability and major neurological impairments. Identifying early symptoms and providing effective treatment to CSM patients before development of irreversible spinal cord damage are critical to maintaining these patients’ quality of life.
The number of patients undergoing surgical treatment for CSM each year has increased 7-fold from 1993 to 2002.4 Approximately 10% of all patients 55 and older display clinical CSM; yet, 85% of adults older than 60 exhibit radiographic cervical spondylosis with risk of progression.1,5 The mean age of CSM patients is 56, and the population of people over the age of 60 in the United States is expected to double by 2050 to nearly 100 million Americans; therefore, it is imperative that physicians remain informed on how to diagnose and manage this condition, as it is likely to become more prevalent in the decades to come.6
Pathophysiology
Although the exact pathophysiology underlying CSM remains uncertain, clinical symptoms generally result from spinal cord compression secondary to a multitude of factors.7 Cervical cord compression can occur as a result of disk herniation, infoldings of ligamentum flavum and facet joint capsule, or canal stenosis.8 Spondylosis, or degeneration of intervertebral discs and joints, may result in compression of the surrounding vascular and neural structures that contribute to disease severity.9 These various mechanisms lead to compression of the spinal cord and subsequent neuronal damage; they are classified as either static or dynamic mechanical compression.10,11 Static risk factors are constant and result in direct injury via stenosis of the cervical canal, and dynamic factors involve repetitive injury. The repetitive injury typically arises from flexion or extension of the cervical spine, which can stretch the axons, making them more susceptible to secondary injury.
Static risk factors include congenital spinal stenosis, herniation of disk material, osteophytosis, and ligamentous hypertrophy. Congenital spinal stenosis narrows the spinal column, which can lead to local ischemia, neural cell injury and apoptosis, and a high likelihood of subsequent development of cervical myelopathy.8,12,13 In addition, discs can herniate and compress the cord or the lateral walls of the annulus fibrosis can tear.14 Both disk herniations and annulus fibrosis tears put stress on the vertebrae and osteophytes can develop. Osteophytic development can stabilize the vertebrae, and overgrowth can also contribute to compression of the cord and surrounding vasculature.8 Ossification of the ligamentum flavum or posterior longitudinal ligament has also been known to narrow the spinal canal and contribute in progressive cervical myelopathy.8
Dynamic mechanical compression results from pronounced movement beyond the typical range of motion that leads to translation and angulation of the column or overlapping of the laminae and buckling of the ligamentum flavum.10,11,15 Some examples include hyperextension of the neck that could lead to a collapsing of the ligamentum flavum dorsally into the spinal canal or anterolisthesis during flexion that squeezes the spinal cord if a compressive lesion is pressing against the cord.8,15
In addition, multiple studies have shown that the histopathologic changes seen in cervical myelopathy are comparable to those observed in isolated spinal cord ischemia.10,16 Vascular flow can be diminished through the anterior spinal artery and the radicular arteries when the vessels are stretched over a disk or vertebral body. Vessel ischemia leads to poor perfusion of the vascular-dependent oligodendrocytes, leading to oligodendrocyte death via apoptosis and subsequent neural demyelination.17 Other factors potentially implicated in the pathophysiology of CSM include impairment of intracellular energy metabolism, free radical-mediated injury, and cation-mediated cell injury.15 The cumulative result of spinal cord damage in CSM is neuronal dysfunction, which causes hallmark symptoms secondary to inhibition of afferent and efferent nerve fibers of the spinal cord, commonly referred to as long-tract signs.18
Natural History
Although cervical myelopathy only develops in a small percentage of patients with spondylosis, its development is frequently insidious in nature.7,18 Age-related degeneration is the primary cause of CSM, however traumatic spinal injuries to the disk can compound the degenerative process in a younger patient.9 Wu et al19 reported that myelopathy may be more common in men than women, especially in those with labor-intensive employment.20 Moreover, the same study reported an overall incidence of CSM-related hospitalization to be 4.04 per 100,000 person-years and specified that the incidence of CSM increases with age. Lees et al21 reported that CSM is associated with a progressive deterioration of motor and sensory function, and 95% of patients who report gradual intervening periods of disease progression and stability, whereas others have demonstrated that the progression of CSM is highly variable, with a portion of patients reporting a benign form of the disease.22 Some risk factors for poor prognosis in those with CSM include degree of disease progression on recognition, age, and the duration of symptoms.1,23
In its severe form, CSM is generally thought of as a condition best managed with surgery. Systematic reviews have shown that 23% to 54% of patients initially managed nonoperatively subsequently undergo surgical treatment.24 Age, symptom duration, and preoperative neurological function are important prognostic indicators of surgical outcome and should be considered to devise the best treatment plan.2
Signs, Symptoms, and Diagnosis
The most common initial symptoms of CSM are subtle and include difficulties with fine hand dexterity and gait instability or falls.7 Sadasivan et al25 found that there was an average delay in diagnosis of CSM of 6.3 years, during which time patients declined by an average of 2 Nurick grades, illustrating the need for prompt diagnosis and treatment of this debilitating disease (Table 1).22
Nurick Scale for Clinical Myelopathy Evaluation (0 to 5 Points)
As in most diseases, the first step in evaluating a patient with suspected CSM is conducting a thorough history and physical examination. Due to compression of the spinal cord and roots from degenerative changes in the spinal column, different symptomatology is seen at the level of compression and below. Patients commonly experience motor deficits from insults to upper and lower motor neurons. Along with motor dysfunction, patients with CSM also experience variable sensory losses from compression of specific sensory spinal tracts, The physical examination findings that all physicians should be mindful of in a patient with suspected CSM are summarized in Table 2.
Symptoms and Physical Exam Findings Commonly Seen in Patients with Cervical Spondylotic Myelopathy
The original symptom severity scale for CSM was published by Nurick in 1972 and was based solely on gait deterioration. This scale has largely been replaced by a more holistic grading system termed the Japanese Orthopaedic Association Myelopathy Evaluation Questionnaire (also known as JOA scale).13,22 This questionnaire incorporates 6 subscores based on upper extremity motor, lower extremity motor, upper extremity sensory, lower extremity sensory, truncal sensory, and bladder function.26 Although the JOA scale has become the preferred grading scale to assess overall patient debilitation, the Nurick scale is still occasionally used to assess the impact of gait dysfunction on activities of daily living.27 The Nurick and JOA symptom severity scales for CSM are shown in Table 1 and 3, respectively.
Japanese Orthopaedic Association Scale for Cervical Myelopathy Evaluation (0 to 17 Points)
Upper Extremity Findings
Upper extremity deficits are common as a result of lower motor neuron insults. At the affected vertebral level, lower motor neuron symptoms predominate, including weakness, muscle atrophy, fasciculation, hyporeflexia, and hypotonia found in the upper extremities.7 In addition, 75% of patients have decreased dexterity in the intrinsic muscles of the hand, leading to problems in handwriting, typing, shirt buttoning, or other fine motor tasks.7,28 Other more specific signs include the classically described “myelopathy hand” by Ono et al,29 which includes positive finger escape sign, grip and release test, and intrinsic hand muscle wasting.30 The hyperactive pectoralis reflex has been shown to be a very sensitive (84.8%) and specific (96.7%) marker of myelopathy at the level of cervical 2 (C2) to C4.31 If the level of compression is at C6, a positive inverted radial reflex may be present.15,32 One final upper extremity reflex that may be elicited in CSM patients is the Hoffman sign. The Hoffman sign has a prevalence of 2% in the general population, although a positive predictive value of 68% and negative predictive value of 70% for CSM make it a useful adjunct in diagnosis.33,34 Sensory changes due to compression of the spinothalamic tract, posterior column, and spinal roots can lead to changes in pain, temperature, proprioception, and general dermatomal sensation.7 Specific sensory findings in the upper extremity and thorax in patients with CSM include neck pain (50%), radicular pain (38%), and a positive Lhermitte sign (27%).35 Examples of some additional examination maneuvers are found in Figure 1.
Select physical examination maneuvers to support a diagnosis of cervical spondylotic myelopathy. (A) Finger escape sign; (B) grip and release test; (C) hyperactive pectoralis reflex; (D) inverted radial reflex; (E) Hoffman sign.
Lower Extremity Findings
In CSM, the lower extremities are impacted by injury to upper motor neurons below the affected vertebral level. It is common to observe upper motor neuron symptoms, which are characterized by weakness, hyperreflexia, and hypertonia.36 Gait dysfunction is seen in 80.3% of patients and is the hallmark of CSM, as reflected by Nurick’s original grading system.22,28 Lower extremity weakness is most frequently seen in the iliopsoas muscle (38.8%), followed by the quadriceps (26.3%).28 This causes characteristic changes in gait, including slower gait speed, decreased step length, longer stride time, and increased step width compared with healthy controls, potentially leading up to quadriplegia.37 Other lower extremity symptoms to evaluate include sustained clonus of the foot, defined as >3 beats in succession. This finding has poor sensitivity (11%) but high specificity (96%) for CSM.33 A positive Babinski reflex may also be seen, which carries up to 100% specificity for CSM, although only 13% sensitivity.38 Sensory disturbances in the lower extremities may include proprioception deficits secondary to posterior spinal column involvement, which may be differentiated by the Romberg test and heel-to-toe walking. As in the upper extremity, changes in pain, temperature, and sensation can be seen as well. CSM patients may also exhibit bladder sphincter tone changes, as seen in 44% of patients.35
Harrop et al39 conducted a retrospective analysis of 103 patients being evaluated for cervical degeneration to determine the utility of various physical examination findings in CSM. They found CSM patients demonstrated the following symptoms: gait abnormality (91%), any hyperreflexia (85%), lower extremity hyperreflexia (81%), upper extremity hyperreflexia (67%), Hoffman sign (83%), and Babinski reflex (44%).
Radiographic Evaluation
Imaging is an important modality to confirm a diagnosis of CSM. Plain radiographs, computed tomography (CT) with or without myelography and magnetic resonance imaging (MRI) can all be used to evaluate spinal canal narrowing and pathologic vertebral changes. As in usual diagnostic progression, plain radiographs are often taken before advanced modalities, as they are less costly, faster, and expose the patient to less radiation. However, if suspicion remains high, MRI is preferred for definitive evaluation due to its noninvasive nature, high resolution, and ability to visualize soft tissues.25,40 The imaging modalities in use for CSM and their utility are summarized in Table 4.
Imaging Modalities Used in the Workup of Cervical Spondylotic Myelopathy with Their Respective Utility and Associated Cost
Anterior-posterior and lateral radiographs may be utilized to determine the spinal alignment and Torg-Pavlov ratio, which has been shown to be lower in patients with CSM than in controls and is demonstrated in Figure 2.42 A ratio of less than 0.8 is defined as stenotic, and a canal diameter of less than 12 mm often has been correlated with cord compression. Although there is the potential that anatomic variability makes the Torg-Pavlov ratio unreliable, a study by Suk et al43 concluded that despite limitations, the Torg-Pavlov ratio is a useful tool in the assessment of cervical spinal stenosis.44 Lateral radiographs may also be used to determine range of motion and maximal flexion, which have recently been found to be associated with myelopathy symptoms by Nicholson et al.45 The 2018 study by this group showed that increased C2 to C7 range of motion and maximal flexion are associated with milder myelopathy symptoms, suggesting CSM patients may compensate for canal stenosis by hyperflexion.45
Lateral cervical spine radiograph demonstrating Torg-Pavlov ratio at C5. Calculated by dividing canal diameter (A) by vertebral body diameter (B). Ratio <0.8 signifies canal stenosis. Canal diameter <12 mm is correlated with cord compression.
CT scans have a variety of uses in the diagnosis of CSM and are the gold standard for diagnosing ossification of the posterior longitudinal ligament, a separate entity causing similar neurologic effects.46⇓–48 CT myelogram can be used as a near-equivalent test in patients with contraindications to MRI. CT films are useful for preoperative evaluation and planning as well as assessment of the transverse foramen of each cervical vertebrae through which the vertebral arteries pass.
MRI is considered the gold standard to confirm the diagnosis of CSM; through changes in signal intensity (SI), it can be used to determine the severity of degeneration and cord compression, quantitate the diameter of the canal, and detect intrinsic cord abnormalities.49 A systematic review of 26 articles by Tetreault et al50 found 3 predictors of negative surgical outcomes based on MRI, including greater number of high SI segments on T2-weighted imaging, combined high SI changes on T2 and low SI changes on T1, and higher SI ratio. Interestingly, there was no association between neurological outcome postsurgery and maximum cord compression ratio, number of discs compressing the cord, or diameter of the cord.
Electrophysiological studies are not routinely used to diagnose cervical myelopathy but may be of use to rule out other conditions, including carpal tunnel syndrome, multiple sclerosis, amyotrophic lateral sclerosis, subacute combined degeneration, or other neurologic diseases that demonstrate characteristic electromyographic patterns. The literature on this subject is inconsistent secondary to cost, availability, and the use of multiple modalities, such as motor-evoked potentials, somatosensory-evoked potentials, nerve conduction studies, electromyography, and intraoperative monitoring.51 Electrophysiology may play isolated roles in diagnosis, follow-up, and treatment.51
Management
Once CSM is diagnosed, the primary decision to be made is whether to initially manage the condition operatively or nonoperatively. CSM is generally considered a surgical disease, as it has been found that with nonoperative treatment the rates of significant activities of daily life impairments are 6% at 1 year, 21% at 2 years, 28% at 3 years, and 56% at 10 years.24
To date, there have been no high-level studies comparing outcomes in operative versus nonoperative management. A prospective, multicenter, nonrandomized trial comparing surgical versus nonoperative treatment of CSM was undertaken by Sampath et al52 in which the authors concluded that surgical patients have better outcomes, including functional status, overall pain, and neurologic symptoms, despite having a greater burden of disease preoperatively. Rhee et al38 published a systematic review on CSM management in 2013 in which they recommended against nonoperative treatment as the primary modality in patients with moderate-to-severe myelopathy. They suggested those with mild myelopathy may be managed nonoperatively but should be followed routinely for deterioration.53
AOSpine North America and the Cervical Spine Research Society (CSRS) recently released a 2017 guideline for management of CSM based on severity.54 For mild CSM, surgical intervention or supervised trial of structured rehabilitation should be offered. If the patient fails to improve or deteriorates on nonoperative management, surgical intervention should be pursued. For patients with moderate-to-severe CSM, they strongly recommended surgical intervention. For patients with evidence of cervical cord compression but lacking signs of myelopathy or root compression, counseling regarding risk of progression, education regarding signs and symptoms, and regular clinical follow-up should be given. Finally, for patients with cervical cord compression and evidence of radiculopathy, the authors suggest offering surgical treatment or structured rehabilitation with close follow-up. The 2017 practice guidelines for management of CSM based on severity are summarized in Table 5.
AOSpine North America and Cervical Spine Research Society Guidelines for Management of Cervical Spondylotic Myelopathy Based on Japanese Orthopaedic Association Severity
Nonoperative Management
To date, there have been no studies to determine the optimal nonoperative treatment modality for CSM, likely due to the varied needs of a diverse patient population and the multifactorial nature of its pathogenesis. Treatment modalities include bed rest, medications, steroids, injections, exercise, soft collar, rigid collar, cervical traction, thermal therapy, and others.53 In the absence of established guidelines, treatment should center around patient symptoms. Some research has shown that rigid collars are unlikely to help and that spinal manipulation has not shown long-term benefit.55 There have been anecdotal reports of manipulation, traction, and massage worsening neurologic symptoms; therefore, these modalities should be avoided.53,56,57 Expense, time, and resources must also be considered when creating a treatment plan.
Operative Management
Surgical management of CSM is undertaken with either an anterior, posterior, or combined approach depending on the specific pathology. Patients with significant kyphosis, 1 to 2 vertebral level involvement, or ossification of the posterior longitudinal ligament are commonly managed via an anterior approach. Those with >3 levels involved, cervical stenosis, posterior compression, or congenital stenosis would likely benefit from a posterior approach.58 The mainstays of surgical procedures performed via a posterior approach consist of laminectomy with or without posterior spinal fusion and laminoplasty.59⇓⇓–62 Procedures via an anterior surgical approach include anterior cervical discectomy and fusion, anterior cervical corpectomy and fusion, or cervical disk arthroplasty.63⇓–65
From a review of 302 surgically managed CSM patients by Fehlings et al,66 overall perioperative and delayed complications rates are 15.6% and 4.4%, respectively. Some of the more common complications are cardiopulmonary issues (3.3%), dysphagia (3.0%), superficial infection (2.3%), pseudarthrosis (1.8%), C5 radiculopathy/palsy (1.7%), worsened myelopathy (1.3%), non-C5 radiculopathy/palsy (1.0%), epidural/wound hematoma (1.0%), durotomy (1.0%), and others, which occur in <1% of cases. In anterior and posterior approaches specifically, minor/major complications are seen at rates of 11.4% and 18.7%, respectively. Wound infection is seen more commonly in posterior approaches (4.7% posterior, 0.6% anterior), C5 radiculopathy/palsy is seen equally in both approaches (1.9% posterior, 1.7% anterior), and dysphagia is slightly more common in anterior (0.9% posterior, 2.3% anterior).
When to Refer to a Specialist
A review of 16 “excellent”-rated studies of surgical treatment of CSM found 3 factors that predict worse outcomes: longer duration of symptoms, advanced age, and more severe preoperative myelopathy.67 Due to the subtle onset of symptoms, known progressive nature, and potential morbidity and mortality of this disease, the primary care physician is vital to the early detection and initiation of appropriate treatment pathways. As patients age, spinal cord compression without signs of myelopathy is seen more frequently. By the age 50, up to 31.6% of the population have MRI evidence of cord compression, and this number rises to 66.8% after 80 years of age.68 A review by Wilson et al.69 showed that only 22.6% of patients with evidence of cord compression will go on to develop myelopathy. For asymptomatic patients with evidence of cord compression, the consensus AOSpine and CSRS recommendations are to reassure, counsel on the risk of progression to CSM and worsening of cord compression, educate on relevant signs and symptoms to be aware of and the importance of reporting them promptly, and regular clinical follow-up without intervention.54,70 Due to the relative difficulty of making a definitive diagnosis by physical examination findings as well as the broad differential of common presenting neurologic complaints, considering referral to a specialist in the setting of any concern for CSM is recommended. Before referral, we recommend that plain radiographs of the cervical spine should be taken as well as MRI if suspicion is high. If imaging does not exhibit signs of CSM, the patient may be referred to a neurologist for further work-up of other differential diagnoses. Early comprehensive diagnostic evaluation and regular follow-up are essential to recognize this disease, slow its progression, and prevent morbidity.
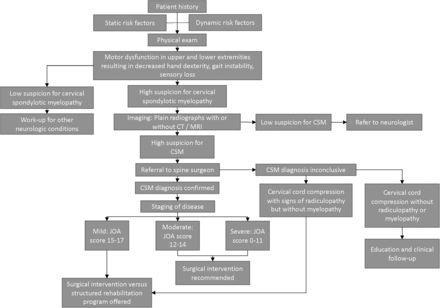
The mainstay of treatment for CSM is surgery; therefore, we recommend immediate referral to a spinal surgeon once a definitive diagnosis of CSM is made. For patients with mild CSM, defined as a JOA score of 15 to 17, the AOSpine/CSRS recommendation is to offer surgical intervention or a structured rehabilitation program.54 These patients should be referred to a specialist spine surgeon in the field of orthopedics or neurosurgery for prompt evaluation for surgical intervention.54,70 Any patient with moderate (JOA, 12 to 14) or severe (JOA, 0 to 11) CSM is recommended to undergo surgical intervention.54 These patients must be referred to a surgeon without delay to prevent further clinical deterioration. An algorithm to aid in the diagnosis, work-up, management, and appropriate referral of a patient with suspected CSM is provided in Figure 3.
Algorithm to aid in the diagnosis, work-up, management, and appropriate referral of a patient with suspected cervical spondylotic myelopathy. Abbreviations: CSM, Client Services Manager; CT, computed tomography; MRI, Magnetic Resonance Imaging; JOA, Japanese Orthopaedic Association.
Conclusion
CSM is a degenerative disease that commonly presents with subtle symptoms that are easily missed by clinicians, often leading to a delay in diagnosis and irreversible neurologic damage. The hallmark signs of this disease are a decrease in fine motor function of the hands as well as gait instability. Due to the well-described progressive neurological dysfunction seen in patients with CSM, the authors strongly recommend an early spine surgery referral with proper imaging for any patient suspected to have this disease for prompt evaluation and appropriate management. This review of the pathophysiology, natural history, diagnosis, and management of CSM may serve as guidance for any physician in the evaluation and triage of a patient suspected to be affected by this condition.
Notes
This article was externally peer reviewed.
To see this article online, please go to: http://jabfm.org/content/33/2/303.full.
Conflict of Interest: The authors, their immediate family, and any research foundation with which they are affiliated did not receive any financial payments or other benefits from any commercial entity related to the subject of this article. There are no relevant disclosures.
Author contributions: All authors significantly contributed to the document and have reviewed the final manuscript.
- Received for publication May 25, 2019.
- Revision received August 23, 2019.
- Accepted for publication August 25, 2019.